Effect of seaweed on equine microbiome composition
1. Introduction to equine microbiome composition and the effect of Seaweed inclusion in the diet
Horses are free-ranging herbivores of grassland environments adapted to eat large quantities of high fibre grasses (Janis, 1976; Bennett, 1980). Nowadays, with the increase of their use in sports and leisure, many horses are housed during long periods of time and fed daily on two or three meals of forages plus concentrate. The forage component of the diet is sometimes characterized by having low-to medium energy concentration and variable levels of fibre and protein (Micol and Martin-Rosset, 1995). This pattern of feed supply is not as expected by the horse’s phylogenetic adaptation to grassland environments (Janis, 1976). These management procedures have important implications on the utilization of nutrients from both concentrate and forage components of the total ration, on the digestive tract in general and the hindgut in particular and, of course, on the health and welfare of the horse (Hill, 2007). More knowledge is needed and therefore we have conducted research in co-operation with one of our suppliers into the effect of feeding seaweed based supplements on the microbiome composition of the horse and the general health status of the horse.
2. How to analyse the horse microbiome?
Workflow and synergistic application of different techniques to study enteral microbiomes. Integrative and synergistic workflow to study equine microbiomes starting with dividing the fresh sample (a) for two general processing’s, microbial diagnostic in terms of culturomics (b-d) [157] and DNA sequencing approaches (e and f) for population analysis (g-i). A broad range of different aerobe and aerobic culture conditions are used to initiate growth for microbial diagnostic (b), followed by rapid species identification by MALDI-tof mass spectrometry (c). Genome sequencing (d) allows (novel) species identification in case MALDI-tof provided no confident result or if resistance- and virulence encoding genes, or other factors are of particular interest within a species. Both information sources allow identification of bacterial species present in the horse microbiota and their growth conditions. The second part of the sample should be stored native at − 80°C until DNA extraction starts for either sequencing of variable regions of 16S/18S rRNA gene (e) allowing characterizing and quantifying taxonomic entities or sequencing of all genomes (metagenome) present in a sample (f). Further bioinformatics include description of richness (g), diversity indices (h), relative abundances (i) and phylogenetics (j). Combination of classical diagnostics on a large scale and different techniques available to generate genomic data enable deep insights into microbiome composition and characteristics
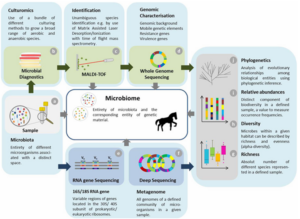
Figure 1. How to asses the composition of the equine microbiome (Kauter et al., 2019)
3.Taxonomic Analysis – Phyla
Before more details about the change in microbiome composition is discussed we need to explain a little bit more about the taxonomic classification of the microbial life in general. In the taxonomy of life we define 3 Domains, Prokaryotes, Archaea and Eukaryotes. Although the Eukaryotes to which humans belong contain a few uni-cellular life forms, most of the microbial life is found in the Prokaryotic and Archaea Domains. The Prokaryotes contain 92 Phyla, whilst the Archaeal Domain contain 26 Phyla.
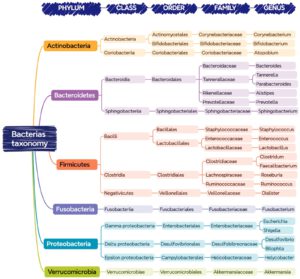
Figure 2. The taxonomic division of microbial life (Martinez-Martinez A.B., 2024)
4. The Equine Microbiome
The Firmicutes are the dominant bacterial phylum of the equine core microbiota, representing around 70% of the total microbiota in the faeces of horses fed a forage-based diet (Venable et al, 2016). This percentage decreased to less than 50% of the total faecal microbiota in horses with conditions such as colitis and laminitis, which result from feeding high starch and or low fibre diets. The percentage of Firmicutes and Bacteroidetes in faecal samples before and after ME supplementation of the 5 horses tested were 54 and 46 and 46 and 44, respectively. The F:B ratio before and after ME was 1.2 and 1.3 , respectively. In addition to a higher F:B ratio in the horses after feeding ME, the microbiome of these horses was more diverse (indicated by Shannon entropy index)
5. Trial design
In a trial, 12 horses in a single stable were being fed a commercial diet, and the object of the trial was to determine the impact of specifically selected seaweeds on those horses after a period of 60 days of being fed the product on top of their standard diet. 5 of the horses were selected for microbiome assay. Faecal samples were taken before the trial and at the end of the trial. Each sample was sent for a 16S microbiome assay. In this result the assay results were aggregated and averaged to provide a “Before Meadow Equine” and an “After Meadow Equine” grouping, with the intention that directional shifts in the relative abundance of key microbiota could be reviewed
6. Taxonomic Analysis in our experiment
At Bacteria Family Level Dougal et al (2014) reported that the Order Clostridiales was found to be represented among the bacteria present across different diet types (high fibre, high oil and high starch). These bacteria were referred to as being part of the ‘core bacteria’ of horses. The Clostridiales were found to be most susceptible to dietary change, with higher relative abundance in the high fibre diet. Within the order Clostridiales are the families Ruminococcaceae and Lachnospiraceae (butyrate-producing bacteria) and Fibrobacteraceae (fibre-degrading bacteria). All three families showed an increase in relative abundance after receiving Meadow Equine for 60 days (See figures below).
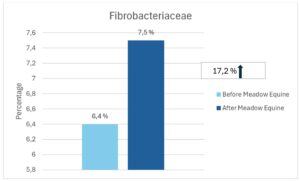
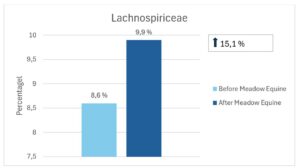
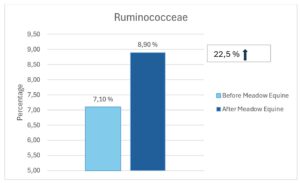
Figure 3. The effect of seawead polysaccharides on the abundance of most important fibre fermenting microbes in horses
Conclusions
Feeding horses a supplement with specially selected seaweeds are resulting in:
1) Improved wellness as a result of diminished subclinical gastric ulcers
2) Better condition due to an improved energy use and an improved hindgut fermentation of forage fibres.
3) Reduced feed costs due to a much better efficiency of nutrient digestion resulting in lower feed consumption
Sources
Bennett D.K . Stripes do not a zebra make. Part 1. A cladistic analysis of Equus. Systematic Zoology 29, 1980 ; pp. 272–287
Janis, C. et al. The evolutionary strategy of the equidae and the origins of rumen and cecal digestion. Evolution, 30, 1976. pp. 757-774
Micol D and Martin-Rosset W. Feeding systems for horses on high forage diets in the temperate zones. In Proceedings of the 4th International Symposium on the Nutrition of Herbivores (ed. M Journet, E Grenet, M-H Farce, M Theriez and C Demarquilly), 11–15 September 1995, Clermont-Ferrand, France, pp. 569–584. INRA editions, Paris
Hill J. Impacts of nutritional technology on feeds offered to horses: a review of effects of processing on voluntary intake, digesta characteristics and feed utilisation. Animal Feed Science and Technology 138,. 2007; pp 92–117.
